Plasma Cell precursor and Other Disorders
Category: Plasma Cell precursor and Other Disorders
Subcutaneous Daratumumab + Bortezomib, Cyclophosphamide, and Dexamethasone in Asian Patients with Newly Diagnosed Light-chain (AL) Amyloidosis: Subgroup Final Analysis of the Phase 3 ANDROMEDA Study
(PA-373) Subcutaneous Daratumumab + Bortezomib, Cyclophosphamide, and Dexamethasone in Asian Patients with Newly Diagnosed Light-chain (AL) Amyloidosis: Subgroup Final Analysis of the Phase 3 ANDROMEDA Study

Kenshi Suzuki
Professor
Japanese Red Cross Medical Center
Introduction: Systemic AL amyloidosis is a rare disorder of clonal CD38+ plasma cells, characterized by deposition of amyloid fibrils leading to tissue damage and organ dysfunction. Subcutaneous daratumumab (DARA SC) is a human CD38-targeting antibody. Combining DARA SC with VCd (bortezomib, cyclophosphamide, dexamethasone) improved outcomes for AL amyloidosis versus VCd alone in the phase 3 ANDROMEDA study. Here, we report a subgroup final analysis of Asian patients in ANDROMEDA.
Methods:
Newly diagnosed AL amyloidosis with measurable hematologic disease, ≥1 involved organ, cardiac stage (Mayo 2004) I-IIIA, eGFR ≥20 mL/min, and no symptomatic MM. Patients were randomized 1:1 to receive DARA SC plus VCd (D-VCd) or VCd. All patients received VCd six 28-day cycles with or without DARA SC. Primary endpoint was overall hematologic complete response (CHR)rate; key secondary endpoints included major organ deterioration progression-free survival (MOD-PFS), survival, and safety.
Results:
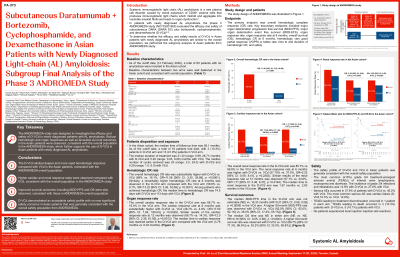
Among 388 randomized patients, 60 were Asian (D-VCd, n=29; VCd, n=31). Baseline characteristics were well balanced between arms and consistent with the overall population. Median follow-up in the Asian subgroup was 59.1 months, and the median treatment duration was 21.3 months for D-VCd and 5.3 months for VCd. The overall CHR rate was 69.0% for D-VCd and 16.1% for VCd (odds ratio, 11.6; 95% CI, 3.4-39.9; P< 0.0001). With longer follow-up time, higher CHR rates were still observed in the D-VCd arm compared with the VCd arm. MOD-PFS favored D-VCd-treated patients (HR 0.21;95% CI, 0.08-0.58). Median OS was not reached in either arm (HR=0.19; 95% CI: 0.05, 0.68; p-value 0.0043). The 60-month survival rate was 89.7% for D-VCd arm and 53.2% for VCd arm.
36 participants (D-VCd: n=15; VCd: n=21) in Asia subgroup were evaluable for cardiac response, with 66.7% overall cardiac response in D-VCd and 19% in VCd respectively. The cardiac response rate at 6-month was 46.7% and 4.8% in D-VCd and VCd groups, which is consistent with overall population results. Median time to cardiac response (D-VCd: 3.8 months, VCd: 6.0 months) was reached earlier in the D-VCd compared with the VCd. 37 participants (D-VCd: n=21; VCd: n=16) in Asia subgroup were evaluable for renal response, with 85.7% overall renal response in D-VCd and 68.8% in VCd, respectively. Median time to renal response (D-VCd: 2.0 months, VCd: 2.8 months) was reached earlier in the D-VCd compared with the VCd.
The safety profile of D-VCd observed was generally consistent with the primary analysis. Asia subgroup showed a generally consistent safety profile with the overall population.
Conclusions:
The addition of DARA SC to VCd was superior to VCd alone in Asian patients, resulting in deeper hematologic response, organ response and improved clinical outcomes, including MOD-PFS and OS, with a safety profile consistent with the overall study population. These data support the use of D-VCd in Asian patients with newly diagnosed AL amyloidosis.
